COVID-19 vaccine is coming to Nevada — and more restrictions?
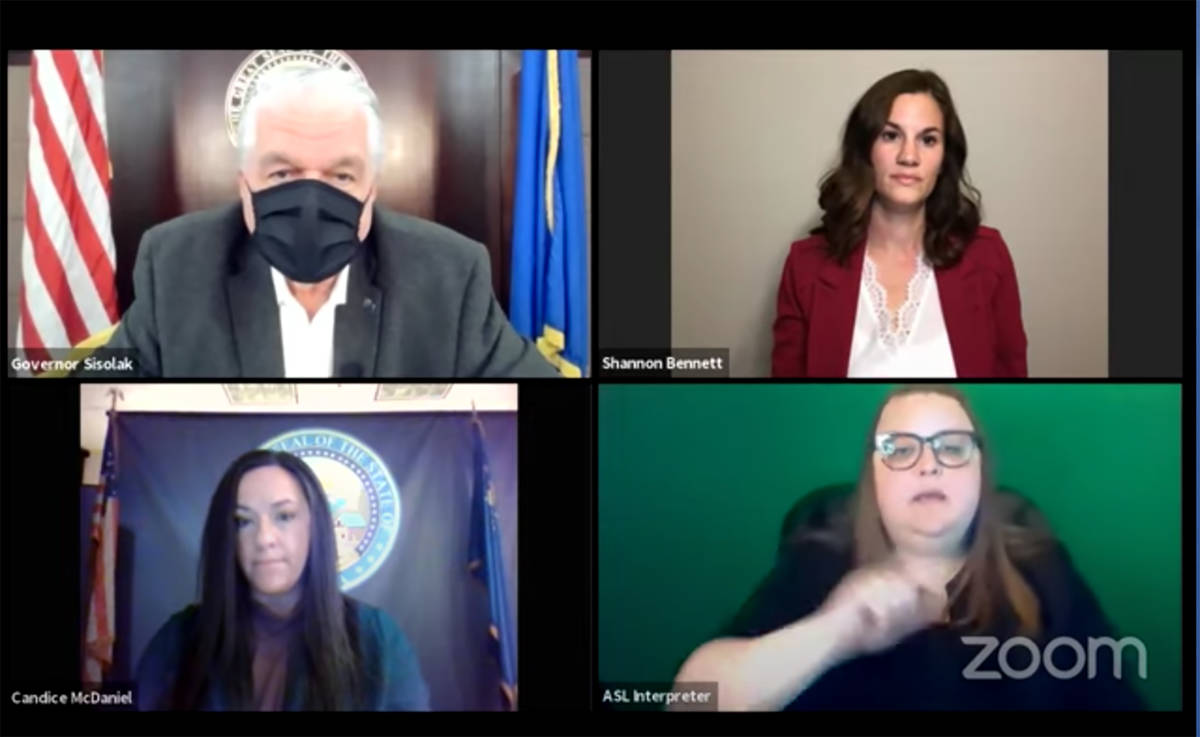
Gov. Steve Sisolak said Wednesday that he expects the first doses of a COVID-19 vaccine to be available in the state in about a week but asked Nevadans to remain vigilant.
“There is hope on the horizon,” Sisolak said during a late afternoon virtual news conference. “There is definitely a light at the end of the tunnel.”
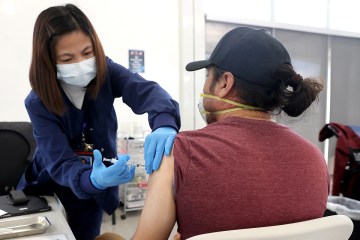
Sisolak was joined by state public health officials who said that the vaccine would first be made available to health care workers at hospitals.
Others near the front of the line for the vaccine include correctional facility staff and members of law enforcement, said Shannon Bennett, immunization program manager with the state Division of Public and Behavioral Health.
Some of the initial doses will go to those who work or live in long-term care facilities and staff from CVS and Walgreens who will be administering the vaccine to them, she said.
Until a vaccine is widely available, Nevadans must take precautions against the virus, such as staying home as much as possible and avoiding large gatherings, the governor said.
“Federal officials have widely reported that we won’t see large-scale vaccination throughout America until the spring,” Sisolak said. “So we need a bridge to get there. Here in Nevada, that bridge between today and the day we have large-scale vaccination must be built by all of us.”
The governor also cautioned that cases are increasing across the state.
“We have a lot of work to do, and it will take all of us to recommit ourselves to turning things around before a vaccine is widely available,” he said.
Nevada reported 35 deaths from the coronavirus on Wednesday, the second-highest single-day increase since the start of the pandemic.
Sisolak also suggested that further restrictions may be on the horizon. On Nov. 22, he restricted the size of gatherings to 50 people and reduced occupancy at casinos, restaurants, bars, gyms and leisure facilities to 25 percent.
“As I mentioned last week, if we do not begin to see a change in our trajectory, and if this crisis continues to get worse, we will be left in the unfortunate position of having to take stronger actions — something I have been desperately trying to avoid,” he said.
But he did not intend to impose any kind of travel ban or restriction at this time.
“It’s a difficult situation,” he said. “You’re walking on the edge of a knife trying to balance the economy and at the same time a health care system that is being burdened heavily.”
First in line
Federal officials have said they expect there will be about 40 million doses of vaccine distributed across the country in December. But state officials did not provide an estimate of how much vaccine initially would be available in Nevada.
Once the first doses arrive, the state expects more vaccine to arrive on a weekly basis, said Candice McDaniel, health bureau chief of the Bureau of Child, Family and Community Wellness.
State officials said that in prioritizing who will receive the vaccine, they were following guidance from a panel that advises the Centers for Disease Control and Prevention, which on Tuesday recommended initial vaccine doses go to health care workers and residents of long-term care facilities.
“Our framework of priority populations begins with those with the highest risk for contracting COVID-19 and those who we are all dependent on to take care of us — our critical infrastructure workforce in hospitals and medical facilities,” Bennett said.
In its revised vaccine playbook submitted to the CDC, Nevada also included the following in the first group or tier to receive the vaccine: emergency medical services personnel, front-line public health workers, laboratory workers, pharmacists, outpatient and home health care providers, national security personnel and state and local emergency operations staff.
There are about 173,000 people in this first tier in the state. To vaccinate the entire tier would require 346,000 doses because each of the early vaccines requires two doses administered several weeks apart.
The second tier includes education and child care staff, college and university faculty, essential retail workers and prison inmates, among others.
The third tier includes those over age 65 and younger people with underlying health conditions. A final tier is for healthy adults.
The vaccine has not yet been tested on children, who generally experience mild illness from the virus.
FDA authorization needed
Before any vaccine can be distributed, it must receive authorization for emergency use from the Food and Drug Administration.
The FDA will be considering two separate vaccines for emergency use authorization this month after clinical trials to evaluate their safety and effectiveness. On Dec. 10, an independent advisory panel to the FDA will evaluate a prospective vaccine developed by Pfizer Inc. and BioNTech. On Dec. 17, it will evaluate a vaccine by Moderna Inc. that was developed with the National Institutes of Health.
Both companies say that clinical trials have shown their vaccines to be more than 94 percent effective with only minor side effects.
Both vaccines rely upon a new technology that uses genetic code to build an immune response in response to the virus.
“I want to stress to the general public that things are looking a lot better,” Sisolak said. “You know, our numbers haven’t been great. I guess everybody understands that. But we have hope now.
“It is our hope that we can get as much of this as we can out to the general public, we’ll build more and more immunity and we can get back to our regular lives.”
Contact Mary Hynes at mhynes@reviewjournal.com or 702-383-0336. Follow @MaryHynes1 on Twitter. Review-Journal web producer Tony Garcia contributed to this report.