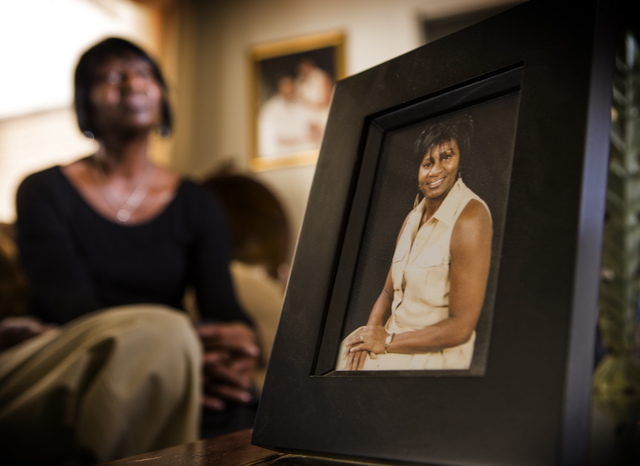Brave risk helps women cheat death
Rosemary Rathbun and Lorrine Rodgers call what’s happened a miracle.
It’s easy to understand why.
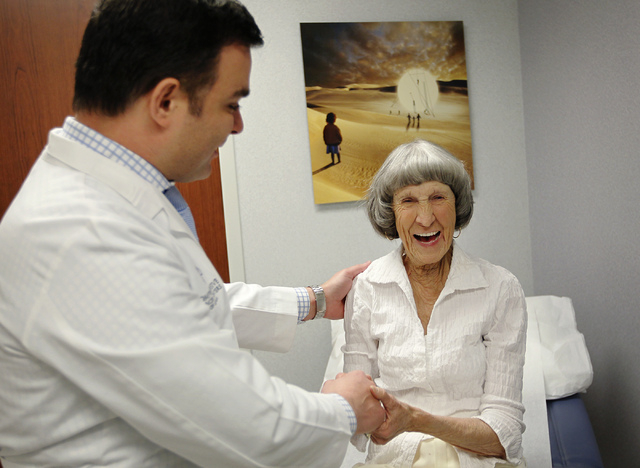
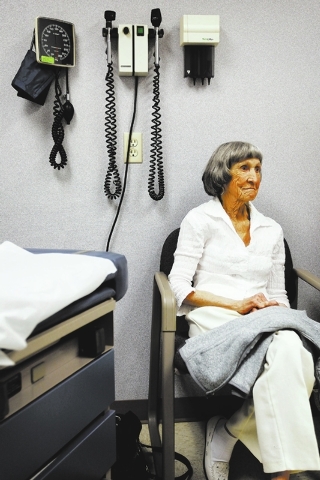
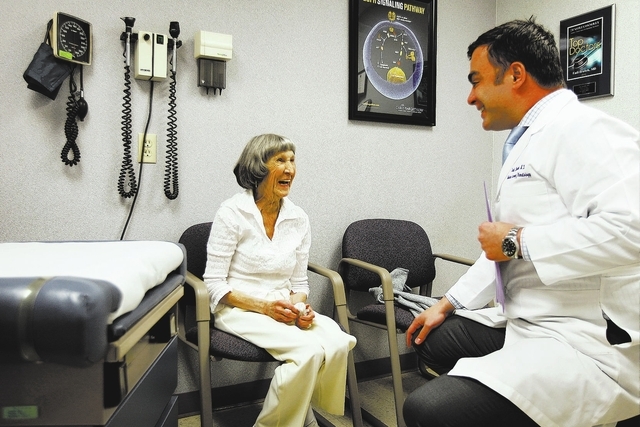
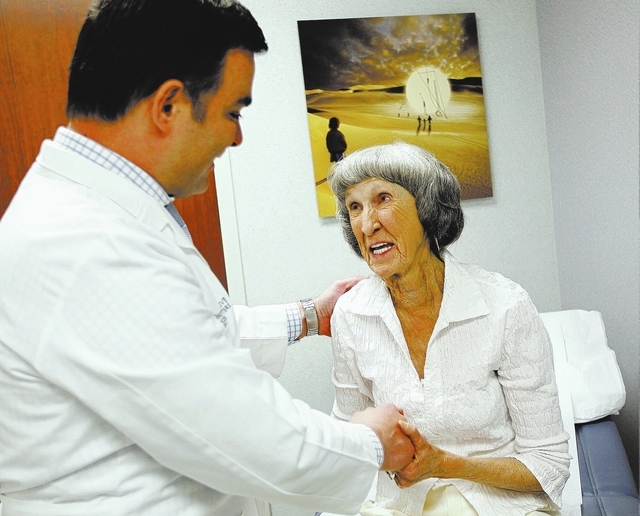
Both Las Vegas grandmothers suffered from cancers that appeared ready to take their lives. Rathbun had been told she should enroll in hospice.
But now, thanks to their participation in the first in-human trial of an antibody drug with the unwieldly code name MPDL3280A, the women are cancer-free.
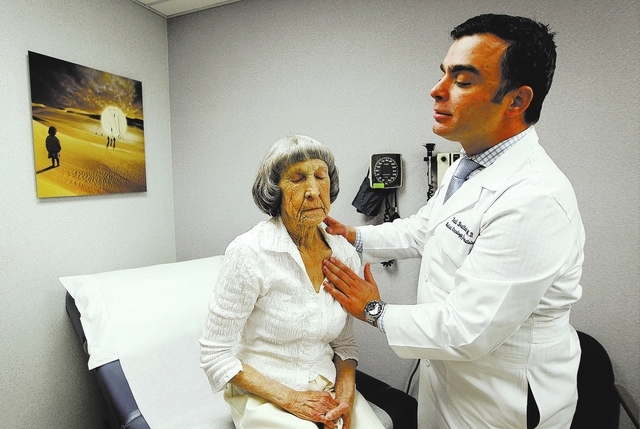
Their oncologist, Dr. Fadi Braiteh, said the new drug unleashed each woman’s own immune system to attack their cancer cells.
“We’re now in a field of cancer treatment known as immunotherapy,” said Braiteh as he sat in an examination room with Rathbun at the Comprehensive Cancer Centers of Nevada Central Valley treatment site on Eastern Avenue. “What’s wonderful about this treatment, what’s so dramatic, is that there don’t appear to be side effects.”
Both the 78-year-old Rathbun and the 56-year-old Rodgers experienced side effects, sometimes severe, from what have been the traditional treatments for cancer: surgery, chemotherapy and radiation.
In July 2009, Rathbun, once a smoker, was diagnosed with throat cancer. Not only did chemotherapy make her “very sick” with nausea and its effects, she said the radiation “fried her throat.”
Even today, said the woman whose cancer later spread to the neck and breast, she’s limited to soft foods.
“For eight months, all I could tolerate was Ensure (nutritional shakes and drinks),” she said.
Rodgers, a hotel housekeeper on the Strip, said the chemotherapy and radiation for her breast cancer caused her to miss three months from work and even more when the cancer spread to her neck. She added that she felt so sickened and weakened by the treatment that she sometimes wonders how she managed to get through a day when she did return to work.
“I couldn’t take off too much time because I’d lose my job,” she said recently as she sat next to her husband in their Las Vegas home.
Given their difficult experiences with what is considered state-of-the-art treatments — losing their hair was the least of their concerns — it isn’t surprising that both women had acute trepidation about trying an unproven therapy with unknown reactions for humans.
They went into uncharted territory, even fearing effects worse than before, because giving into death without using one more possible weapon to stave it off wasn’t attractive to them or their families.
Taking the step put them on an emotional roller coaster — they were riddled with fear one minute, filled with hope the next.
“I did a lot of holding of Lorrine then,” Nelson Rodgers said of his wife. “There were a lot of tears. It was a frightening time.”
To the women’s amazement, the therapy, delivered through an IV for a couple of hours once every three weeks, didn’t sicken them.
“I could go to work without a problem,” Lorrine Rodgers said.
After just six treatments of a 16-treatment regimen, scans already show Rodgers free of cancer.
Rathbun’s yearlong experience with the drug beginning in 2012 was just as easy.
“I couldn’t believe it,” she said. “I went for a few times and the cancer left me.”
Braiteh sounds as amazed as his patients about the drug trial that’s been going on in Southern Nevada and at 17 others sites across the world for about a year.
“It’s incredible,” he said. “Now we have to find out why this therapy works so well on some people and not on others.”
About half of the 15 people on the phase 1 drug trial in Southern Nevada, Braiteh said, have experienced some positive results, which means tumors shrank or cancers did not progress, or, as in Rodgers and Rathbun’s case, cancer disappeared.
“I also had some bladder cancer patients who are now cancer-free,” he said.
Whether his cancer-free patients stay that way is unknown, Braiteh said.
“That is a question we don’t have the answer to.”
Results in Las Vegas have been more positive than those experienced at Yale Cancer Center in New Haven, Conn., the drug’s lead trial site.
A 2013 study of 140 patients there found that 21 percent experienced significant tumor shrinkage. The highest number of positive responses were in patients with lung cancer and melanoma.
What has frustrated researchers for decades is cancer cells’ ability to evade the immune system. Why, researchers wondered, did white blood cells in the lab always kill cancer cells but not in the body?
In recent years, scientists not only learned the answer — cancerous tumors wrap themselves in an invisible molecular shield or camouflage — they also learned that certain drugs could break through the shield.
If the immune system is free to do its job, what researchers have long seen in laboratory Petri dishes — white blood cells attacking cancer cells — can occur in the body, too.
To date, several drugs have been tested to free the immune system. Studies have shown their effectiveness in treating several types of cancers, including breast cancer, melanoma, ovarian cancer and cancers of the head and neck, colon and stomach, but not the prostate.
MPDL3280A, manufactured by Roche Genentech, is designed to prevent a cancer cell’s overexpressed PD-L1 protein from putting the immune system to sleep. By blocking the interaction between PD-L1 and the immune system, the drug boosts a patient’s anti-cancer immune response.
“With this approach, we can take the cancer out from behind the camouflage — that’s what PD-L1 is — and we’re not using anything toxic,” Braiteh said. “That’s what is so brilliant about this approach. We’re just letting the immune system do its job.”
Braiteh couldn’t be happier that cancer fighters now have weapons that don’t produce what he calls “collateral damage.”
“Cancer is a cell of the body that goes nuts,” he explained. “It is what a zombie or vampire is to a human being. And unfortunately what kills cancer cells also kills normal cells. Chemo is like napalm.”
MPDL3280A works best, Braiteh said, on patients who had tumors that had high numbers of cancer cells expressing PD-L1.
“We test people for PD-L1 to see if they should be part of the trial,” he said, adding that the drug trial’s phase 1 continues to recruit patients. Those wishing to join the research study should phone 702-952-3400.
Braiteh said breast cancer patients with the triple negative form of the disease, which Rodgers had, have benefited in the trial.
Although both patients and doctors understandably want progress to quicken for cancer treatment drugs, Braiteh noted the first Federal Drug Administration-approved drug came in 1949.
“Medical oncology is a young field, ” he said.
Exciting to researchers studying MPDL3280A, Braiteh said, is its efficacy in dealing with lung cancer, which has been extremely difficult to treat and becomes incurable once it has spread to other parts of the body.
In September, Europeans were so excited about the results of trials among lung cancer patients — 26 percent of patients responded to treatment — that the head of the European Cancer Congress, professor Cornelis van de Velde said: “Hundreds of millions of euros have been spent chasing the dream of immunotherapy for lung cancer patients, but with zero results.
“These early findings on the effect of the anti-PD-L1 monoclonal antibody, MPDL3280A, suggest that it has the potential to open new therapeutic approaches, particularly for smokers and former smokers.”
Braiteh said a phase 2 study of the drug, focusing on people with lung cancer, is underway in Las Vegas. Five people are being treated with the drug.
“Lung cancer has been a particularly difficult cancer for researchers to deal with,” he said.
Any kind of cancer, Rathbun said, is hard for patients to deal with.
“I’ll never forget when my doctor said I should get into hospice,” she recalled, weeping. “You don’t know what it’s like to be told your life is over. And then along comes this experimental drug. And now I have hope. When you’re a cancer patient, hope is all you’ve got.”
Reporter Paul Harasim can be reached at pharasim@reviewjournal.com or 702-387-2908.