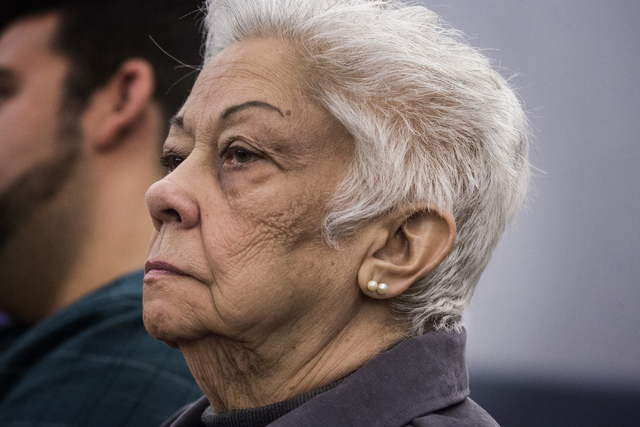Diabetes drug maker downplayed cancer risk, warning label expert testifies in Las Vegas
Like thousands of others, a pair of Clark County women received no adequate warning that the diabetes drug they took could lead to bladder cancer, a warning label expert testified Monday in a potentially multibillion-dollar lawsuit against the maker of Actos.
Takeda Pharmaceuticals, the Japanese maker of the drug, knew about a potential link to bladder cancer for years, but downplayed that risk in packaging inserts, Robert Cunitz, a human factors psychologist from Maryland, told jurors.
Delores Cipriano, 81, of Henderson and Bertha Triana, 80, of Las Vegas filed product liability lawsuits against Takeda last year, alleging they developed bladder cancer after taking the prescription drug pioglitazone, known as Actos.
The cases were combined and the trial, presided over by District Judge Kerry Earley, started last month and is expected to last into May. The women are among thousands across the country who have sued Takeda.
Testimony resumed Monday after a two-week break. Cipriano’s attorney, Robert Eglet, has said he plans to seek a multibillion-dollar verdict, the largest ever awarded in Nevada.
Meanwhile, a federal trial involving hundreds of cases is being heard in Louisiana. A jury on Monday awarded $1.5 million in compensatory damages, according to Bloomberg. The jury will also consider a request for punitive damages.
What the drug company mentioned about carcinogenics in Actos essentially amounted to an “anti-warning,” Cunitz said.
Cunitz read through a series of Actos package insert warnings dating back to 1999, when the Food and Drug Administration approved the drug, and found “there was no warning provided, matching any of the criteria we have. It wasn’t there.”
Cunitz testified under the assumption that prolonged use of Actos could lead to bladder cancer, and said “you don’t have to have perfectly established science to provide a warning about a potential risk.”
He said a drug company should issue warnings as soon as there is “a suspicion of harm.”
In a 2003 email read in court, Takeda executives acknowledged that altering its warning sheet could have “an impact on sales” and it “would be relatively easy for the competition to establish seeds of doubt. Takeda representatives would have difficulty managing these safety concerns.”
The FDA announced in 2011 that taking the diabetes drug for more than one year could be linked to an increased risk of bladder cancer. In July of that year, Takeda altered its warning labels to reflect the cancer danger.
Triana took Actos for two years, between 2005 and 2007, and Cipriano took it for 14 months before the drug maker mentioned the cancer risk in package warnings.
They were both diagnosed with bladder cancer in 2012, and both have had multiple surgeries to remove bladder tumors, according to their attorneys.
Cunitz said the women could not have protected themselves from the risks of the drug because “information about bladder cancer was not presented in a way to attract attention to itself.”
Cunitz told jurors that drug label warnings should attract attention to themselves, be delivered in a timely manner and provide an instruction to avoid harm.
“These warnings were not presented and information about bladder cancer was not presented in a way to attract attention to itself,” Cunitz said. “There was no adequate and effective warning.”
On cross examination, Cunitz acknowledged that he had never spoken to doctors who prescribed Actos to the women.
Cunitz has not worked for a pharmaceutical company or written a drug-label warning himself.
Defense attorneys earlier in the day filed a motion for a mistrial based on a Review-Journal weekend story about judiciary races that referred to Eglet and Earley and mentioned the Actos trial.
Earley said she had not read the article and planned to rule on the motion Wednesday.
The trial is slated to resume at 11 a.m. Tuesday.
Contact reporter David Ferrara at dferrara@reviewjournal.com or 702-387-5290. Follow @randompoker on Twitter.