Las Vegas man 1st in world to use new experimental cancer drug — VIDEO
Spend a few minutes with Anthony Brasich and it quickly becomes clear that he’s a special kind of guy. Nice. Friendly. Great sense of humor, with a fondness for classic one-liners.
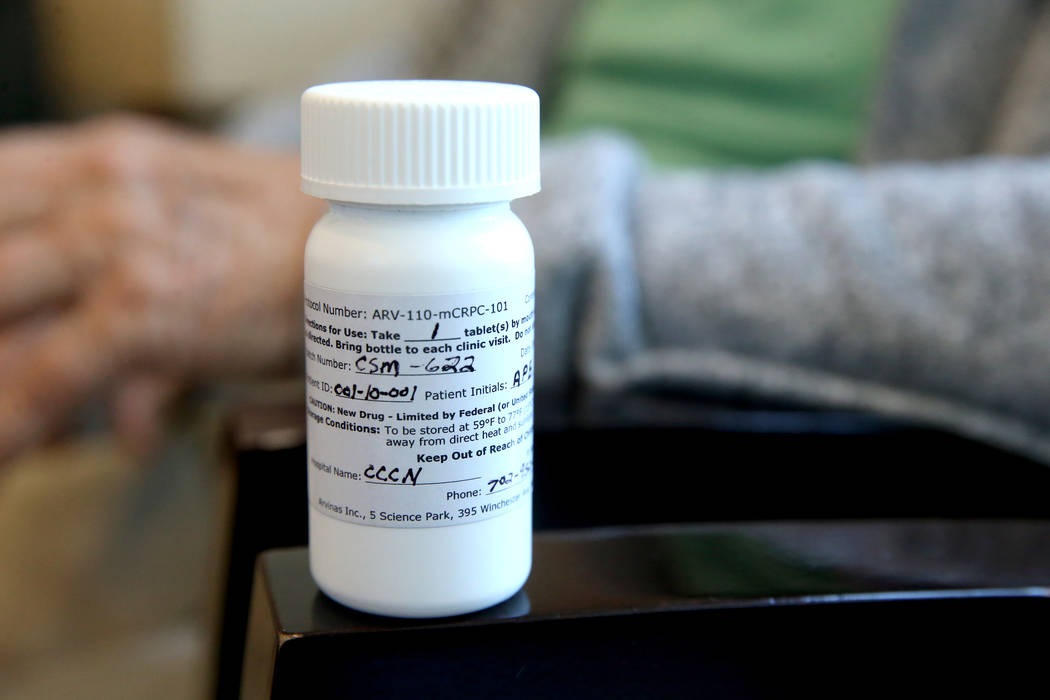
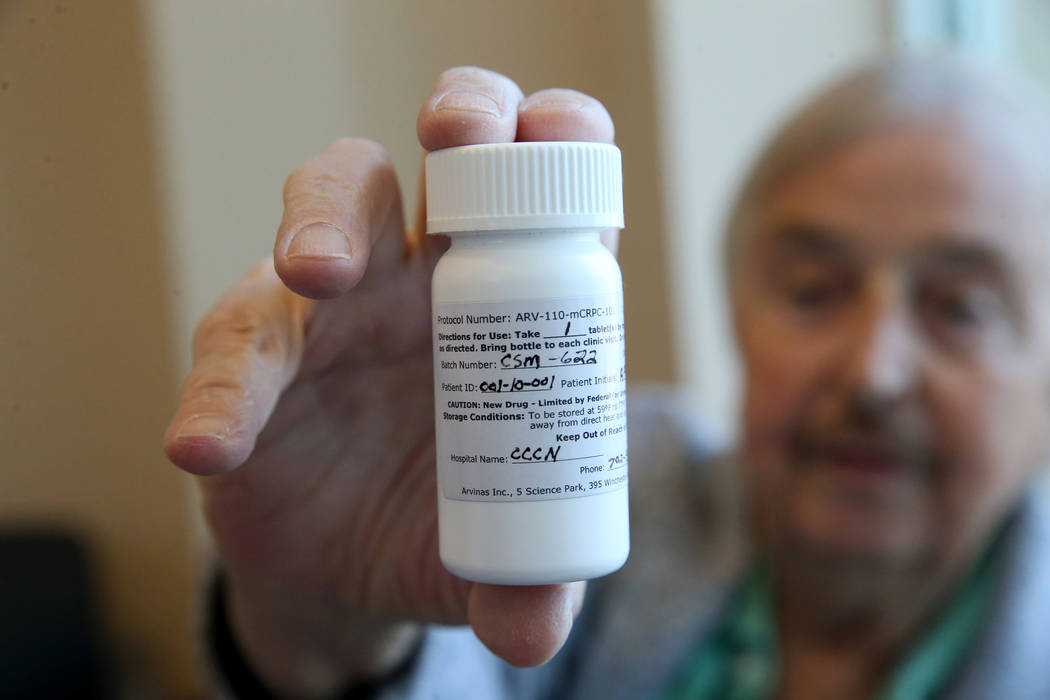
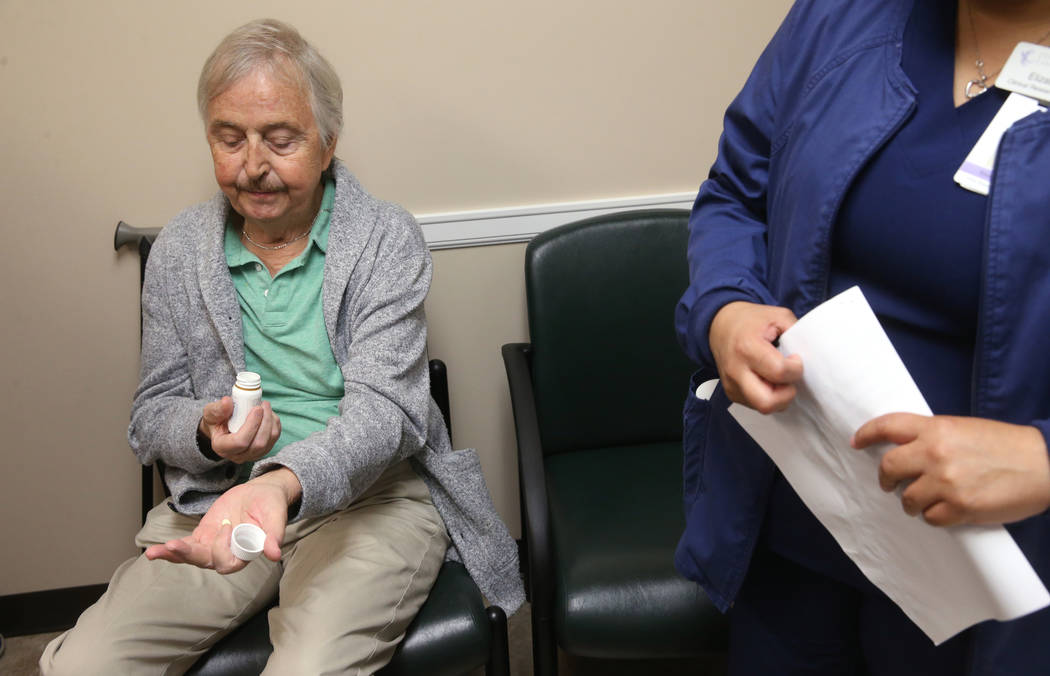
But if anyone needs proof that Brasich, of Las Vegas, really is special, just look at the odd-looking label on the pill bottle that holds his latest once-a-day drug, which designates him as patient 001.
“I’m the one,” Brasich says, laughing. “Hopefully there’ll be another million.”
Brasich, 71, was diagnosed about 10 years ago with stage 4 metastatic prostate cancer and given just a few months to live. That singular designation on the pill bottle indicates that he’s the first patient in the world to participate in phase one clinical trials of an experimental drug called ARV-110.
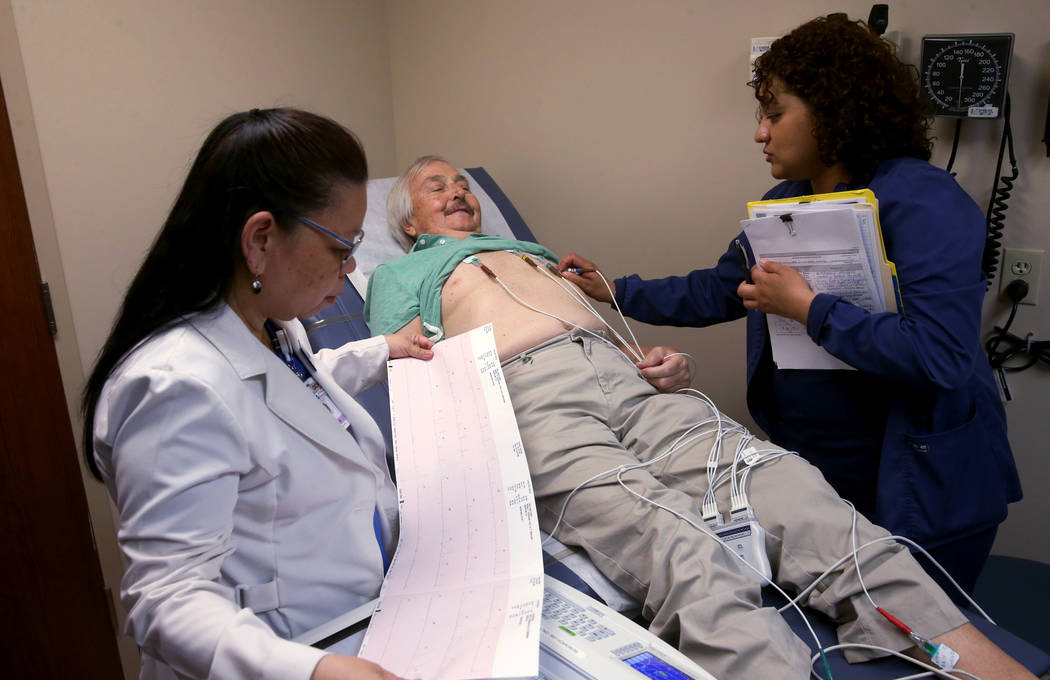
Brasich, a retired casino dealer, has undergone several treatments for cancer over the past decade. Many worked for a while and then didn’t.
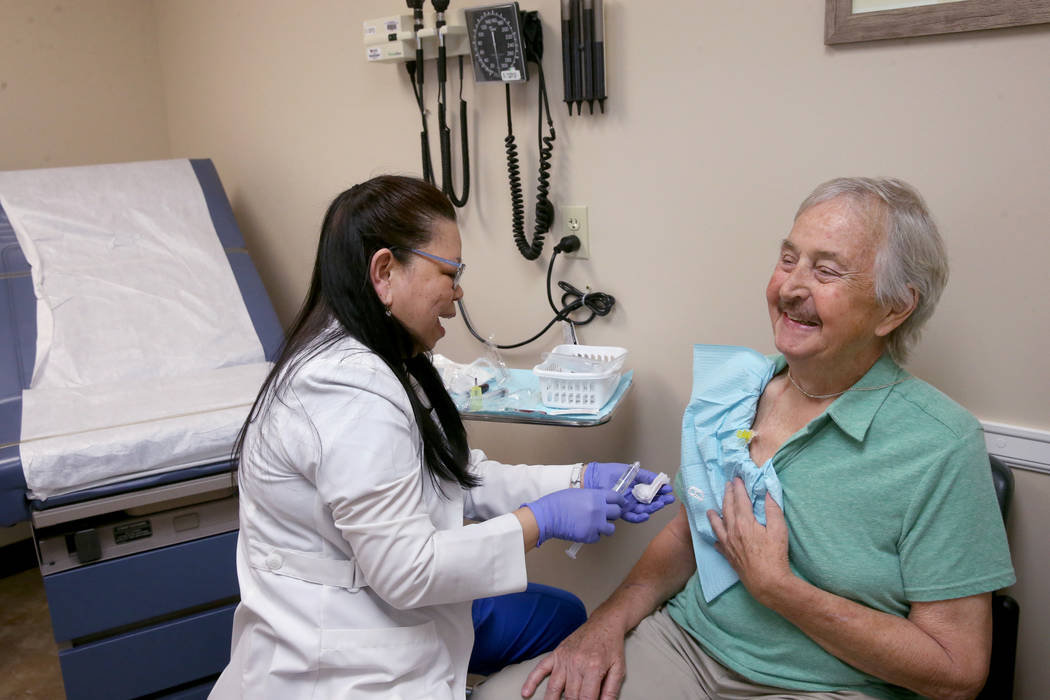
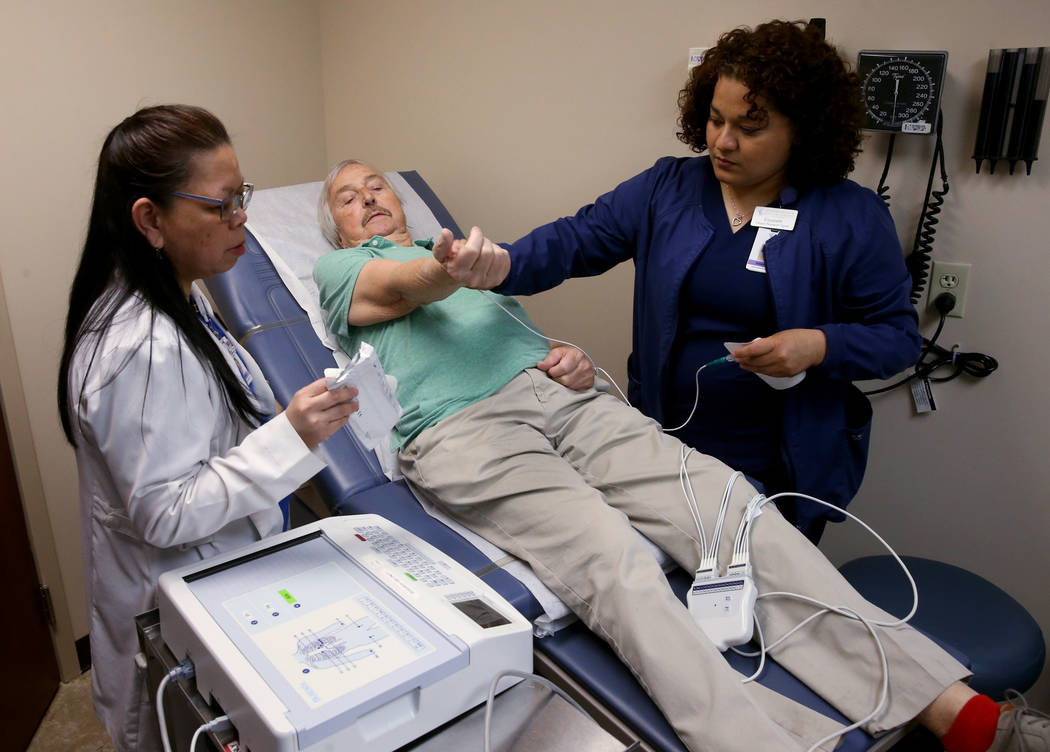
Two-and-a-half months ago, Brasich started taking ARV-110 in the form of a daily pill. He’s tolerating it well, feels reduced pain, and even learned during a checkup Thursday that his PSA level has stabilized. (High PSA, or prostate-specific antigen, levels may signal cancer.)
“Definitely, I feel much better than I had just a couple of months ago,” Brasich says. “On the other drug, I was getting pains in my side and my head, and I couldn’t walk and had blurred vision, and now I’m fine.”
Brasich’s physician, Dr. Nicholas Vogelzang, head of medical oncology at Comprehensive Cancer Centers of Nevada, says prostate cancer needs androgens — male hormones — to grow.
ARV-110 destroys what is known as the androgen receptor, slowing progress of the cancer.
Brasich has undergone chemotherapy, radiation and other drug therapies, as well as a number of experimental treatments. When Vogelzang told him about the ARV-110 clinical trial, “he says, ‘If you want to give it a try, it’s hasn’t been used on humans before.’ I said, ‘Would I be considered a human?’ ”
“I think a rat named Cecil was the first one to use it, and now me,” he jokes.
Actually, Brasich says, “I was very anxious to do it (because) I’m in a position where everything else was failing.”
If all goes well, the ARV-110 clinical trials will continue with the progressive addition of more prostate cancer patients. Then, the study will move into phases that examine the drug’s effectiveness and how it compares with existing prostate cancer treatments.
Even in ideal circumstances, approval of the drug as a treatment option could be two to four years away, Vogelzang says. But he’s pleased with what he has seen so far in Brasich.
A drop in Brasich’s PSA level is “a remarkable thing already,” he says, “so we are very encouraged by these first patients.”
Brasich is thankful that the drug so far seems to be helping him, and says it’d be great if participating in the study led to a cure for his cancer.
But, he says, “moreso, I think, it’s (about) helping other people.”
“I’m not thinking, like, ‘OK, this will work to get me well.’ Fine, if it works like that. But I’d rather see something in the future come up out of this whole thing.
“I think about how I’m the first one,” Brasich says, “and it brings tears to my eyes.”
Contact John Przybys at jprzybys@reviewjournal.com or 702-383-0280. Follow @JJPrzybys on Twitter.