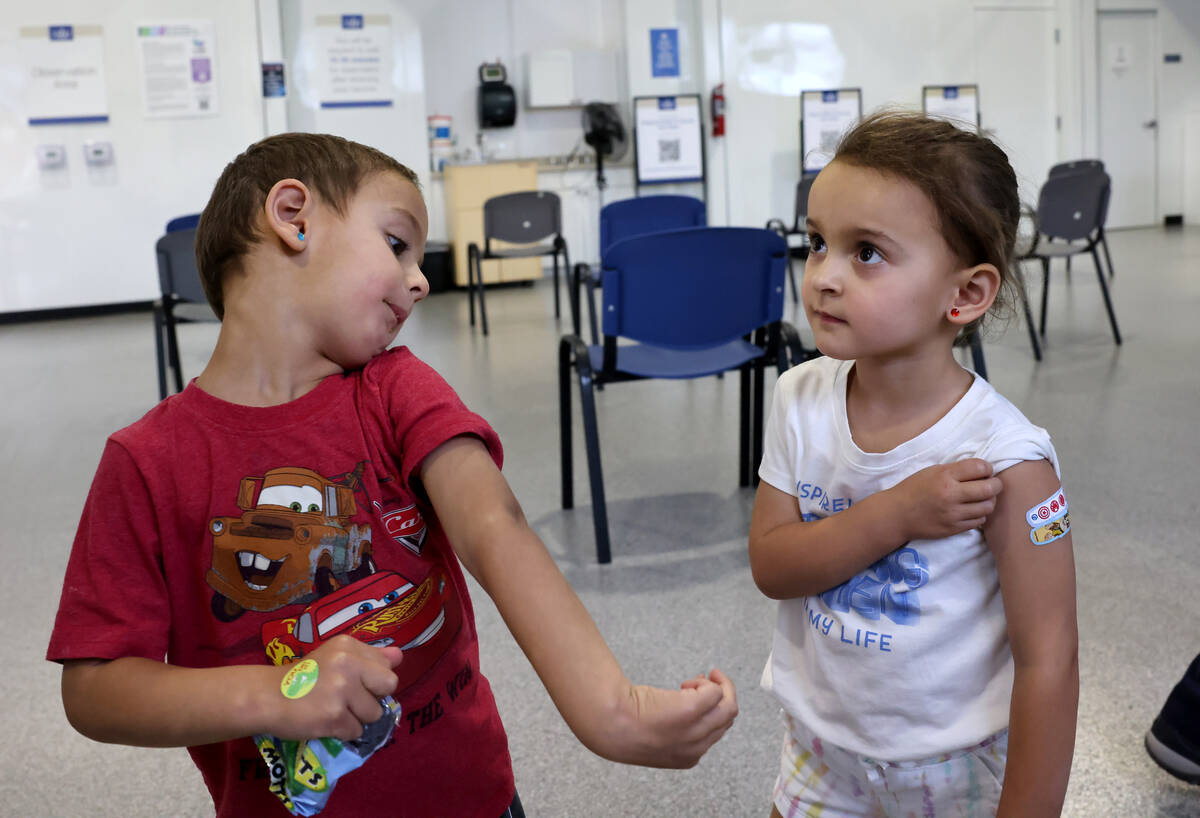
Mission accomplished? What does the end of the COVID emergency mean?
The U.S. government on Thursday will lift the COVID-19 public health emergency that has been in effect for more than three years.
By ending the emergency phase of the public health response, “That kind of puts a period at the end of the pandemic sentence,” Vanderbilt University’s Dr. William Schaffner, an expert on infectious diseases, said.
“We’re clearly past the pandemic phase of COVID-19 here in the United States,” he said. “We no longer have a stressed out health care system. We no longer have as many people becoming sick, seriously ill and dying of this vicious infection.”
However, he cautioned against thinking it’s “mission accomplished.”
“This virus is still with us,” and killing 200 to 300 people in the U.S. every day, he said.
The phase of the disease has gone from pandemic to endemic, or consistently present.
“And in order to keep ourselves protected against this virus on a long-term basis — because it’s not going to disappear — we’re going to have to learn how to keep our guard up,” he said.
Keeping our guard up will include ongoing COVID-19 vaccinations and boosters, treatments and testing, he said.
How will these core components of the public health response be changing as of Thursday?
Vaccinations
Currently, all COVID-19 vaccines are widely available and free of charge, regardless of health insurance or immigration status, according to Shannon Litz, a representative of the Nevada Department of Health and Human Services.
“This will continue until the federally purchased supplies have been depleted,” Litz said, which could be by late summer or early fall.
Once that happens, the vaccines will be covered by both public and private insurance and will remain available to uninsured adults through a U.S. Department of Health and Human Services bridge program, Litz said in an email.
Many Americans will continue to pay zero out-of-pocket for COVID-19 vaccines, according to the federal agency. Vaccines recommended by the Advisory Committee on Immunization Practices (of which Schaffner is a member) are a preventive health service under most private plans and will be fully covered without a co-pay.
COVID-19 vaccinations are currently covered under Medicare Part B without a co-pay or cost-sharing, and this will continue.
Medicaid will continue to cover all COVID-19 vaccinations without a co-pay or cost sharing through September 2024, and will cover what the immunization advisory committee recommends after that.
“The most significant change that the public may experience is the request for insured individuals to provide their insurance card at time of vaccination as vaccinators will be able to charge their insurance for both the cost of the vaccine and a vaccine administration fee,” Litz said.
To find a vaccine location, visit vaccines.gov.
Treatments
Access to COVID-19 treatments such as Paxlovid and Lagevrio generally will not be affected, according to the federal health department.
Out-of-pocket expenses for certain treatments may change, depending on an individual’s medical insurance coverage.
Medicaid programs will continue to cover COVID-19 treatments without cost sharing through September 2024. After that, coverage and cost sharing may vary by state.
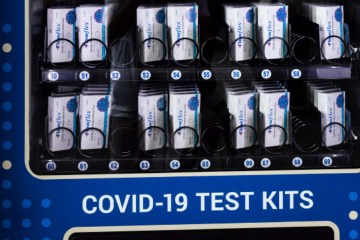
COVID-19 testing
The era of “free” COVID-19 tests is coming to a close — although, of course, they were being funded by taxpayers, but without out-of-pocket expenses.
The requirement for private insurance companies to cover COVID-19 tests without cost-sharing – for both over-the-counter tests and laboratory tests – will end.
Medicare beneficiaries enrolled in Part B will continue to have coverage without cost-sharing for laboratory COVID-19 tests when ordered by a provider. However, there will be out-of-pocket costs for over-the-counter tests.
State Medicaid programs must provide coverage without cost-sharing for COVID-19 testing through September 2024, after which coverage may vary by state.
Depending on the supply and resources, the U.S. government may continue to distribute free COVID-19 tests from the Strategic National Stockpile through the United States Postal Service, states and community partners, according to the federal health department.
Telehealth
The nation embraced telehealth during the pandemic.
Expanded telehealth for Medicare beneficiaries, which was due to expire, will remain in effect through December 2024 as a result of the bipartisan Consolidated Appropriations Act.
Most private insurers already covered telemedicine before the pandemic, according to the Kaiser Family Foundation.
The Drug Enforcement Agency has temporarily extended a public health emergency provision that allows prescribing controlled substances such as Adderall and oxycodone in a telehealth visit, rather than requiring an in-person visit. The length of the extension remains uncertain.
‘This virus is not gone’
On Friday, the World Health Organization echoed the U.S. government’s message by saying that COVID-19 no longer qualifies as a global health emergency.
“One of the things that the WHO is not changing is global surveillance, which continues to be active 24/7/366 looking for new variants of the virus,” Schaffner said. “Which lets you know that all of us are out there recognizing that this virus is not gone, and it could mutate into another variant. …
“We wouldn’t be doing that if we thought it were just ‘mission accomplished,’” he said.
Contact Mary Hynes at mhynes@reviewjournal.com or 702-383-0336. Follow @MaryHynes1 on Twitter.